- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager, Clinical Basics: Monitor
A communication log is a comprehensive record of all interactions between the sponsor and the site that occurred during a study. You may contact the site, for example, to coordinate logistics for an upcoming monitoring visit. And the site may contact you with, for example, questions about the protocol procedures.
It is important to record all emails, phone calls, text messages, etc. to maintain compliance with regulatory requirements and Good Clinical Practice.
There are two ways you can record site communications:
- Email them to your Vault
- Record them manually
Email Communications to Your Vault
To email communications to your Vault:
-
Send an email to: studycommunications@urldomain.veevavault.com, replacing urldomain with the URL domain of your Vault. (Find your URL domain in the URL you used to access Veeva Clinical Basics.)
-
Navigate to Study Management > Site Communications and click the ingested email link.
-
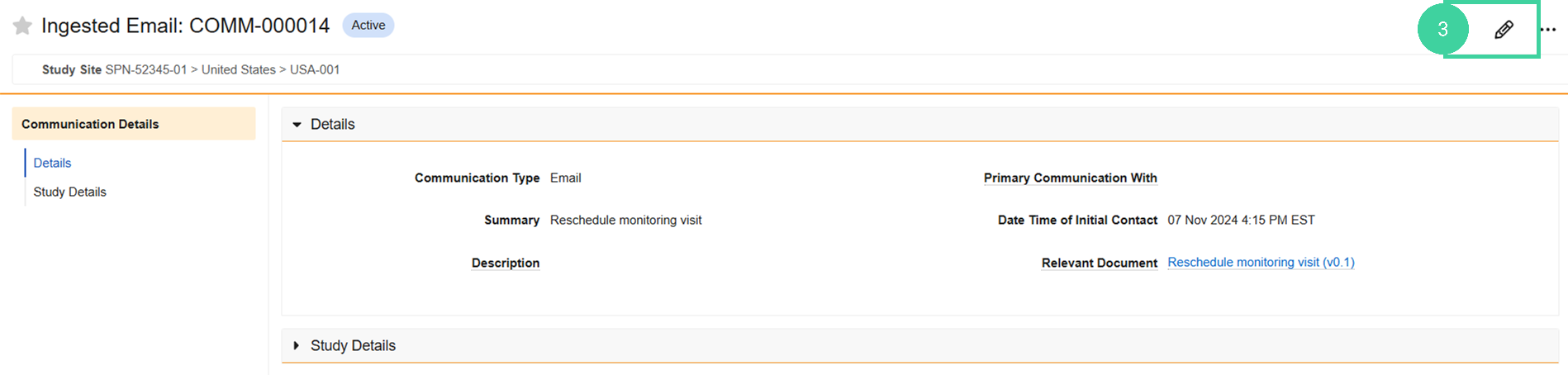
Click Edit.
-
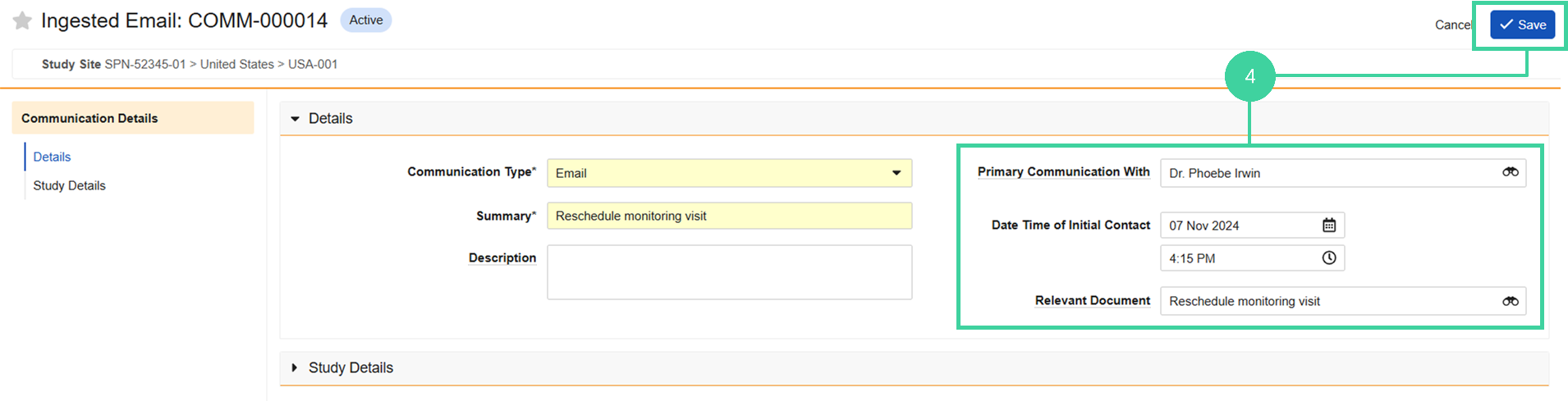
Add the communication details and click Save.
-
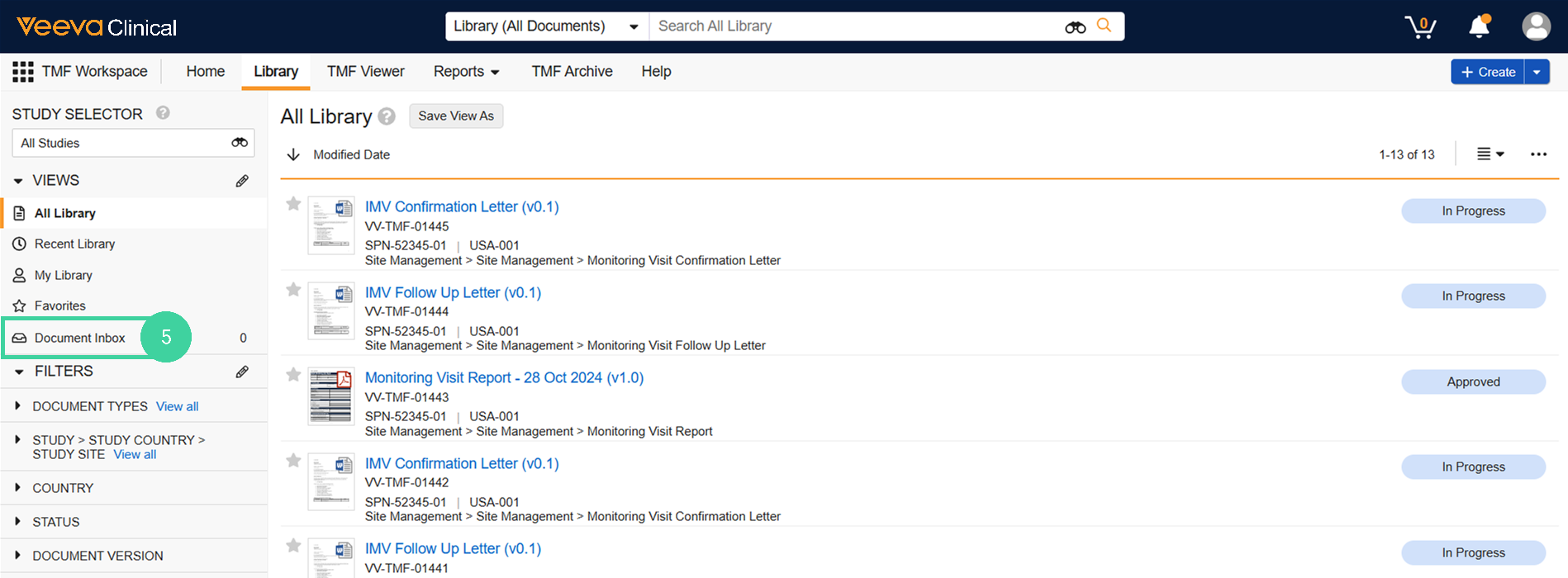
If the ingested email had an attachment, you can find it in the Document Inbox (TMF Workspace > Library > Document Inbox).
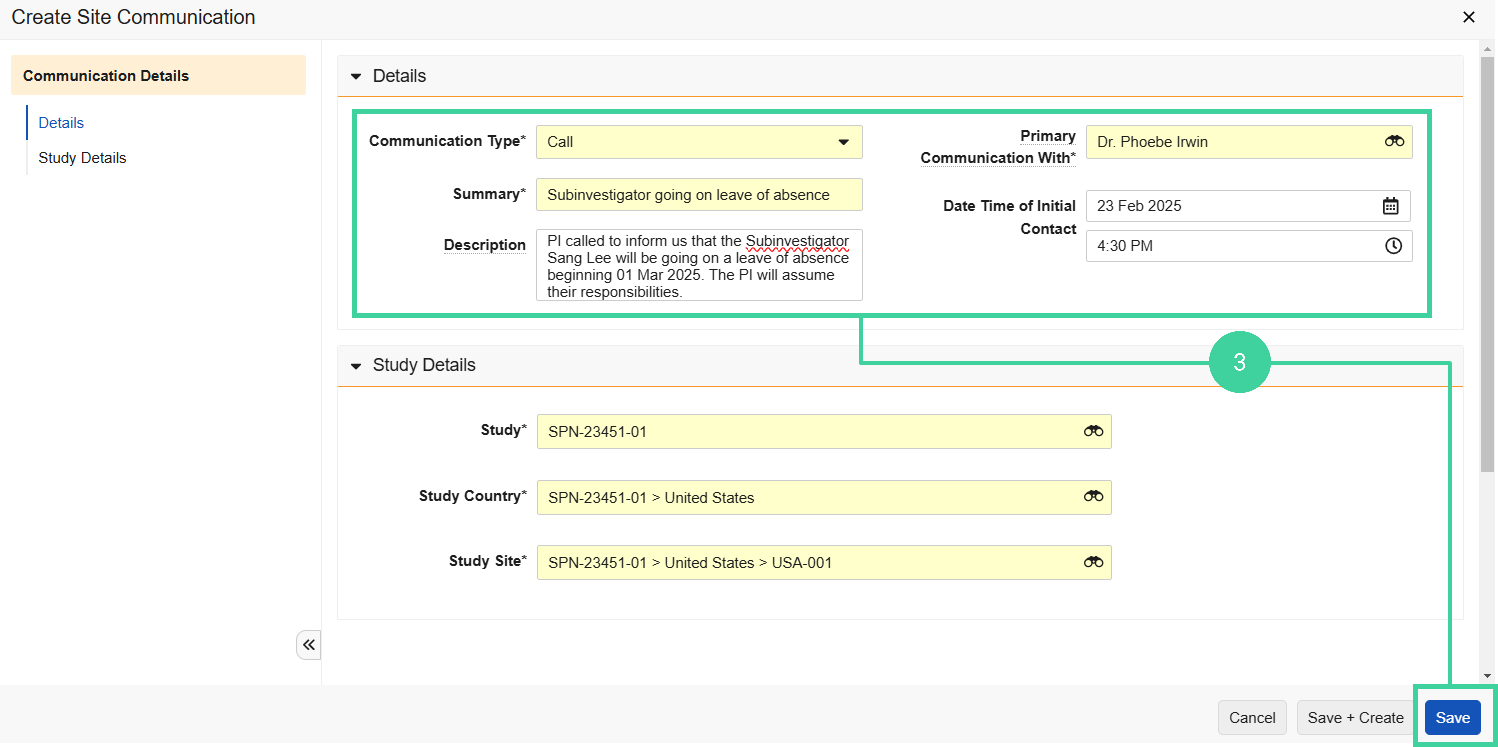
Record Site Communications Manually
To record a site communication manually: