- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager, Clinical Basics: Monitor
If you have not yet completed monitoring visit details, answered visit questions, and recorded any necessary issues and follow up items, do that first.
Once you have completed a visit, you can send it out for review and approval, typically to a Study Manager or Lead Monitor. Once sent, the monitoring visit status will change to In Review.
To send a visit for review and approval:
-
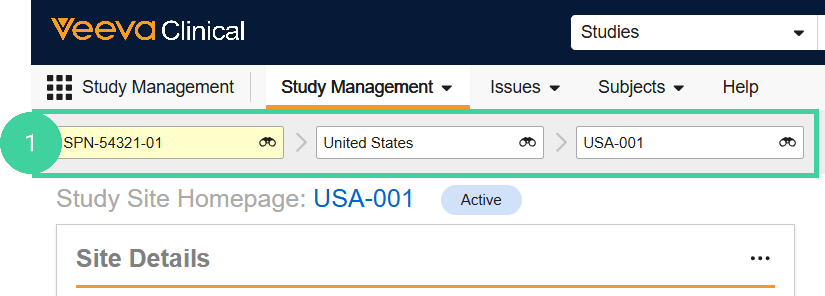
Navigate to the Study Management homepage. Use the study selector to navigate to the correct country and site.
-
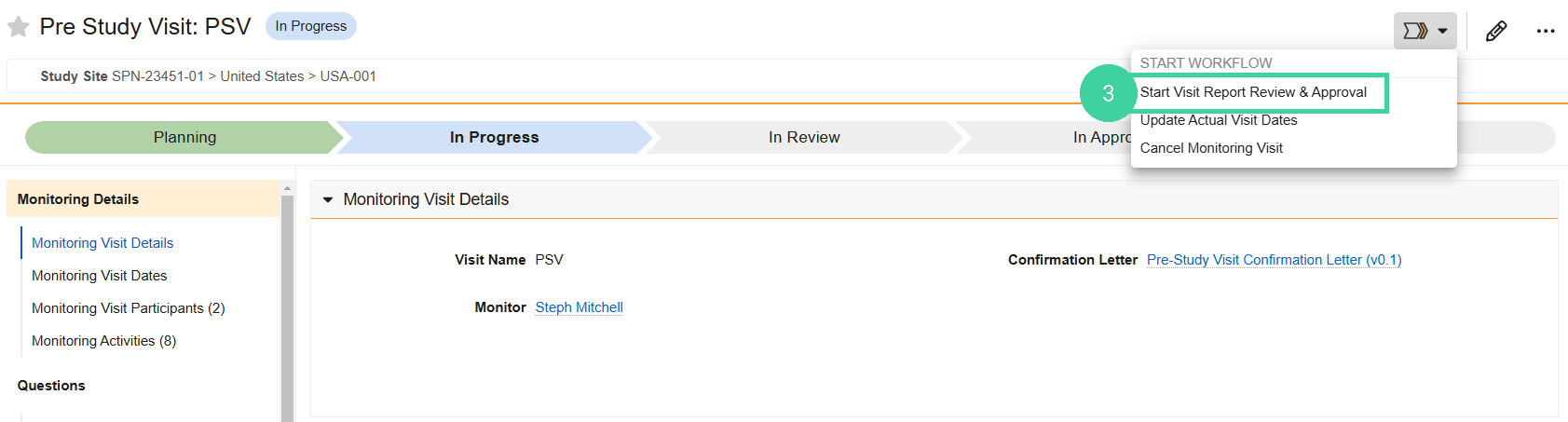
In the Monitoring Plan section, click the Visit you are conducting.
-
Select Start Visit Report Review & Approval from the Workflow Actions menu.
-
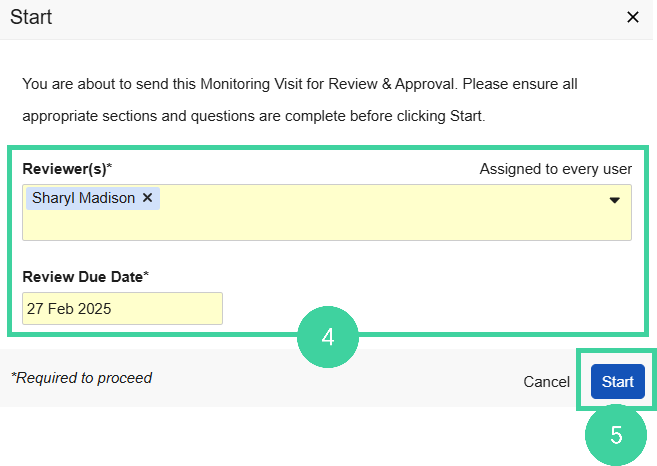
Select one or more Reviewers and Due Date for the review.
-
Click Start. The Visit Report is generated and creates a review task for all reviewers.
What is the next step?