- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager, Clinical Basics: Monitor
If you have not yet started the monitoring visit and completed monitoring visit details, do that first. While you are conducting your visit, you can fill out the monitoring visit as you go.
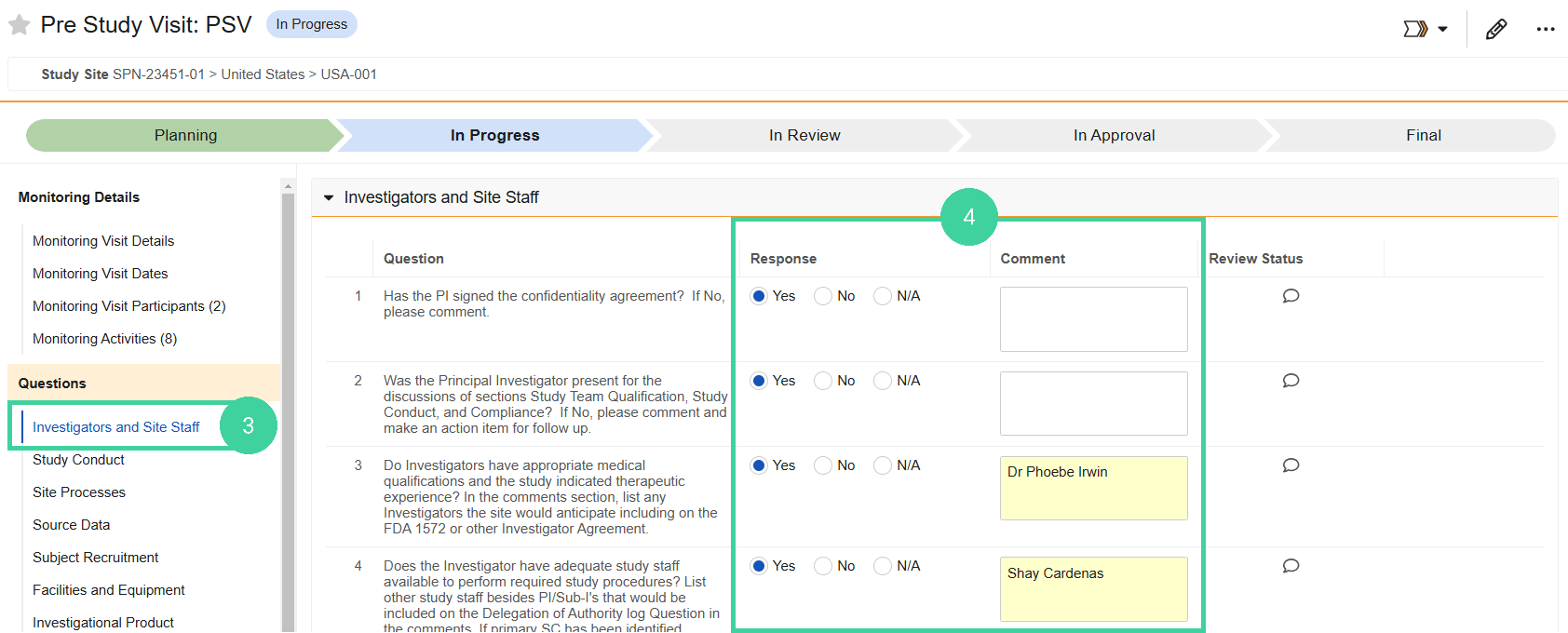
Each monitoring visit type has a specific set of questions, organized into question groups, for you to answer during the visit.
Some questions require you to enter free text comments. Make sure to communicate clearly so that when your Lead Monitor or Study Manager reviews the Visit Report, they will have all the information needed to approve the visit.
To answer visit questions:
-
Navigate to the Study Management homepage. Use the study selector to navigate to the correct country and site.
-
In the Monitoring Plan section, click the Visit you are conducting.
-
Click the first question group from the Questions group on the left. (The name of the first question group will vary, depending on the type of visit.)
-
For each question, answer Yes, No, or N/A and add Comments where appropriate or required. Complete all question groups.
What is the next step?
Review Subject Visits & Enrollment
Record Issues
Record Follow Up Items