- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager, Clinical Basics: Monitor
If you have not yet started the monitoring visit, do that first. While you are conducting your visit, you can fill out the monitoring visit as you go.
To review and complete monitoring details:
-
Navigate to the Study Management homepage. Use the study selector to navigate to the correct country and site.
-
In the Monitoring Plan section, click the Visit you are conducting.
-
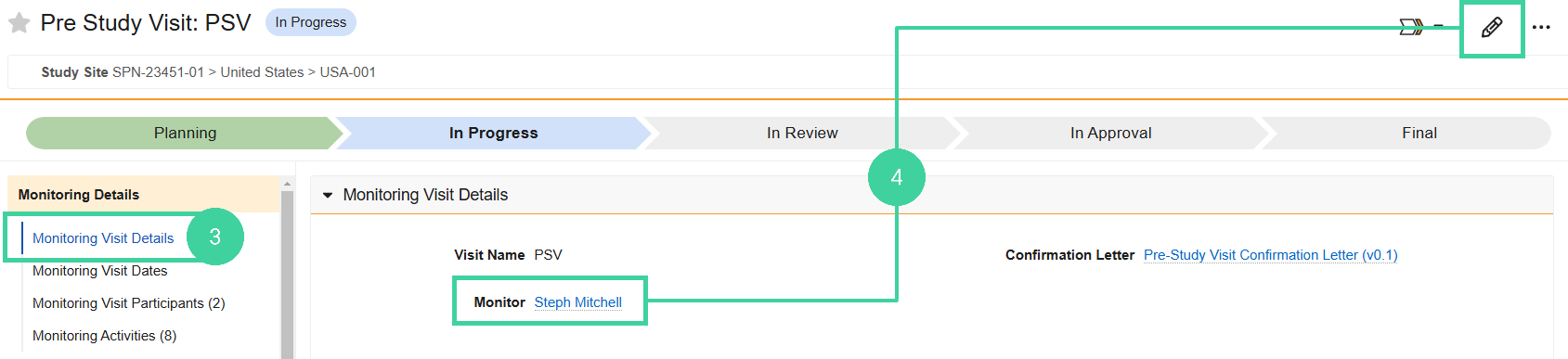
Click Monitoring Visit Details from the Monitoring Details group on the left.
-
Make sure the correct monitor is listed. If not, click Edit and specify the correct monitor.
-
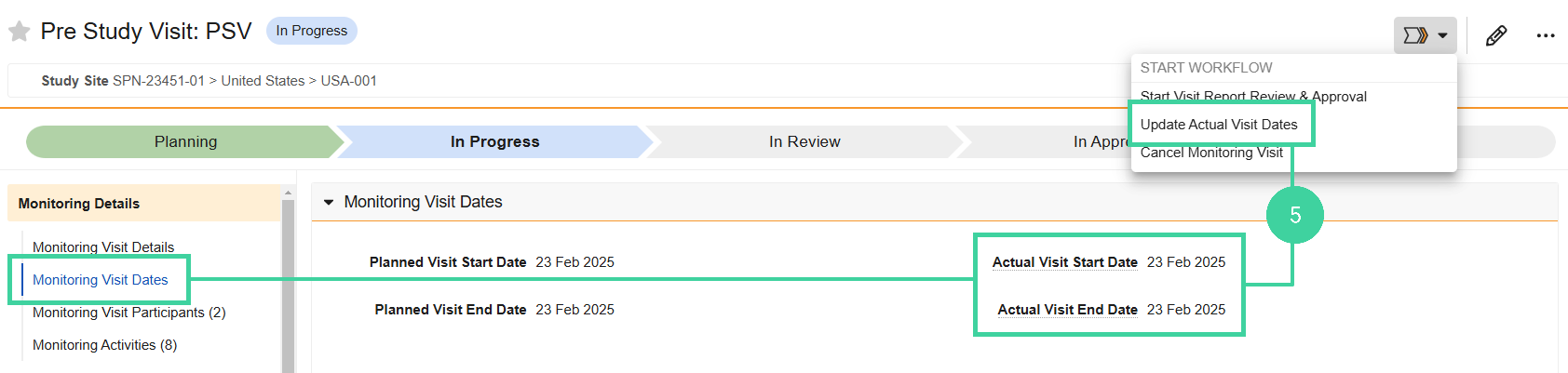
Open the Monitoring Visit Dates section and make sure the correct visit dates are listed. If not, select Update Actual Visit Dates from the Workflow Actions menu and correct the dates.
-
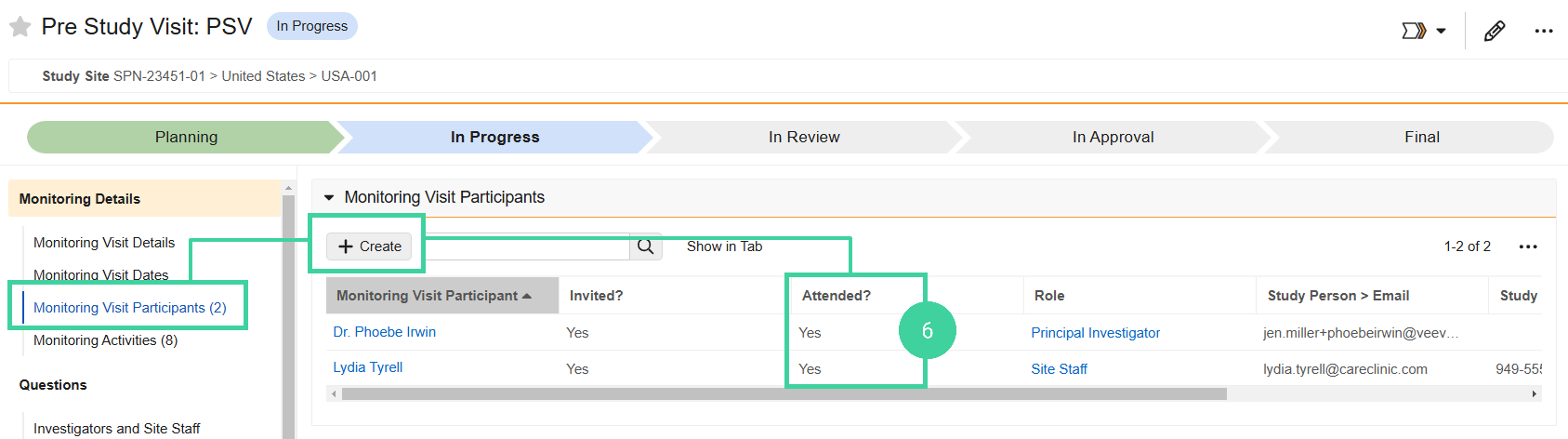
Open the Monitoring Visit Participants section and set Attended? to Yes for everyone who was in attendance. Click Create to add additional participants, if needed.
-
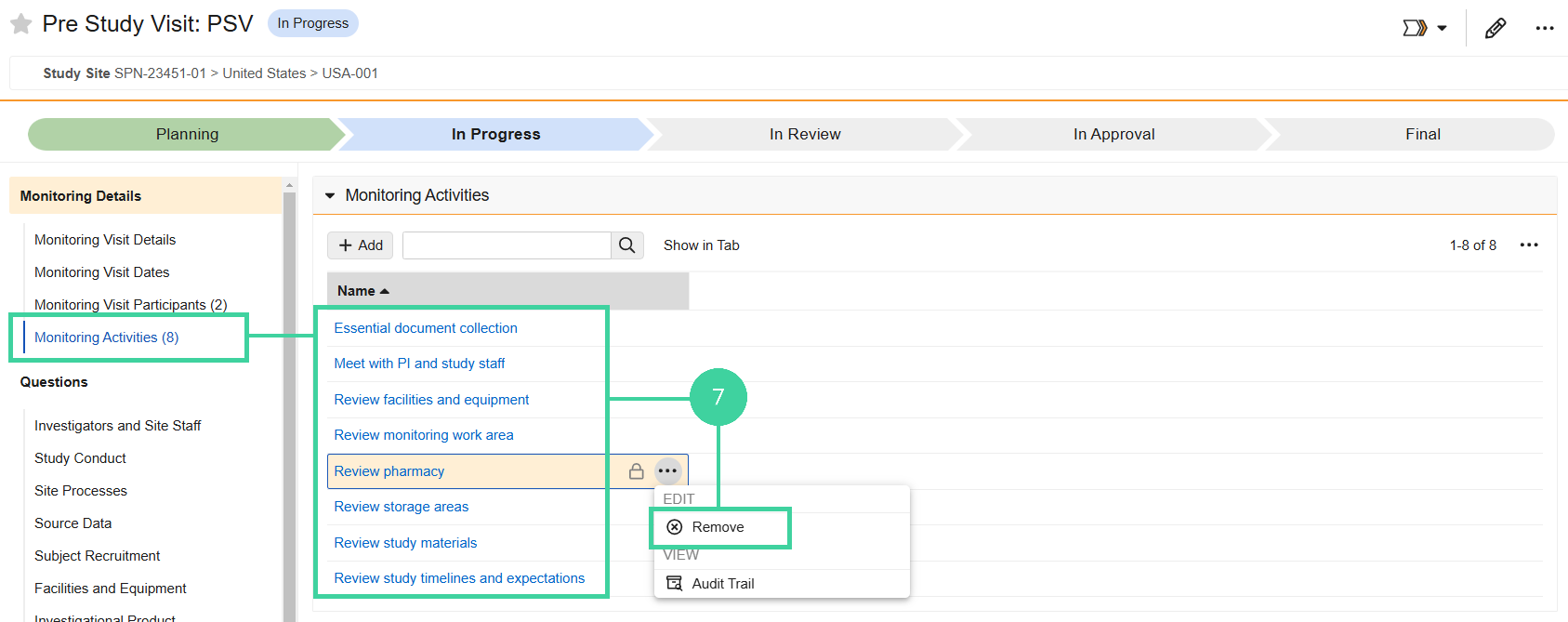
Open the Monitoring Activities section. Hover over any activities that did not occur at the visit and select Remove from the All Actions menu.
What is the next step?