- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager, Clinical Basics: Monitor
Monitoring Visits help ensure a study is conducted ethically and in compliance with regulations.
Veeva Clinical Basics supports the creation and conduct of Pre-Study, Site Initiation, Interim Monitoring, and Close Out Visits.
Create a Monitoring Visit
Creating a visit sets its status to Planning.
To create a monitoring visit:
-
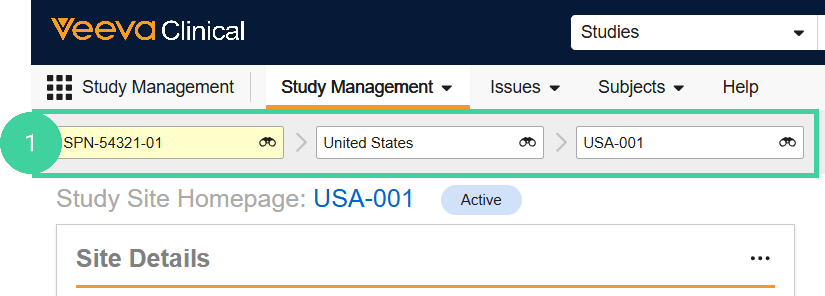
Navigate to the Study Management homepage. Use the study selector to navigate to the correct country and site.
-
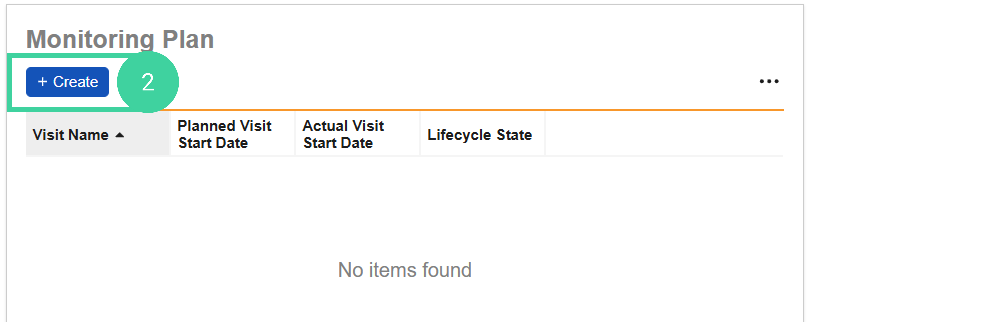
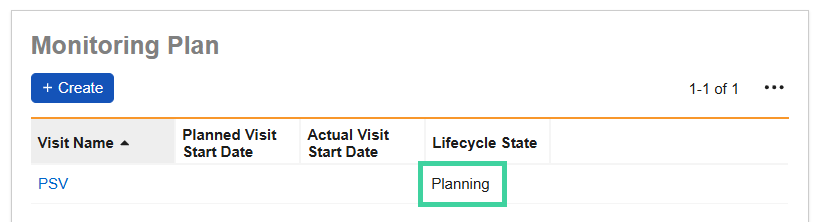
In the Monitoring Plan section, click Create.
-
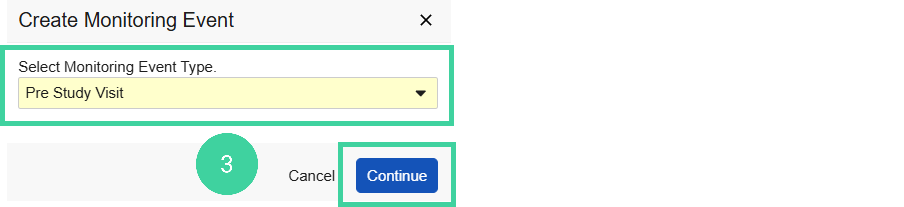
Select the Visit Type and click Continue.
-
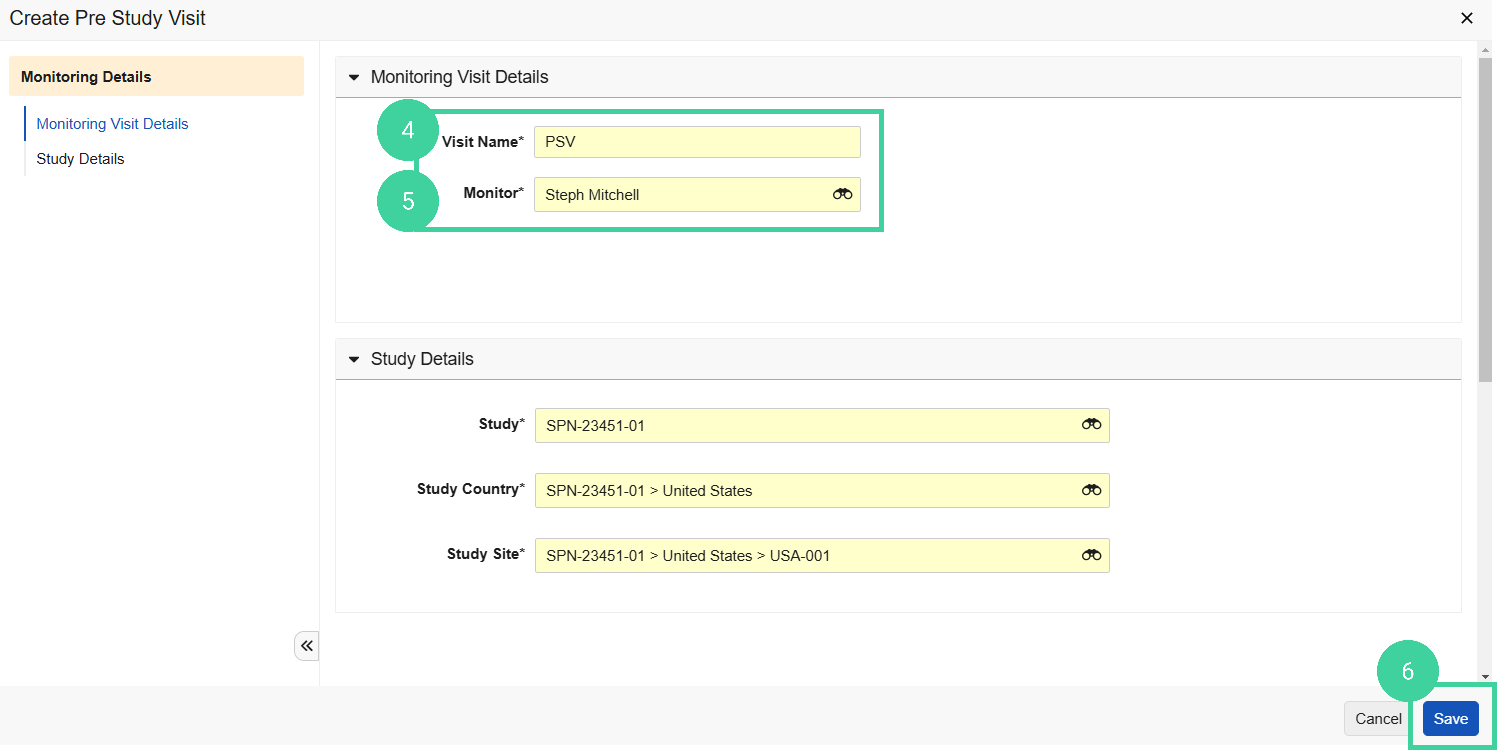
Enter a Visit Name.
-
Select the Monitor who will be attending the visit and click Save.
The status of the visit is Planning.
Confirm a Monitoring Visit
When you know the visit will take place, you can confirm the visit. Confirming a visit creates and classifies a Visit Confirmation letter in eTMF. When ready, this letter should be sent to the Principal Investigator.
Confirming a visit changes its status to Confirmed.
To confirm a monitoring visit:
-
Navigate to the Study Management homepage. Use the study selector to navigate to the correct country and site.
-
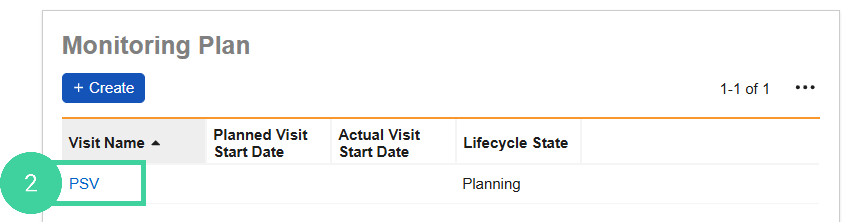
In the Monitoring Plan section, click the Visit you want to confirm.
-
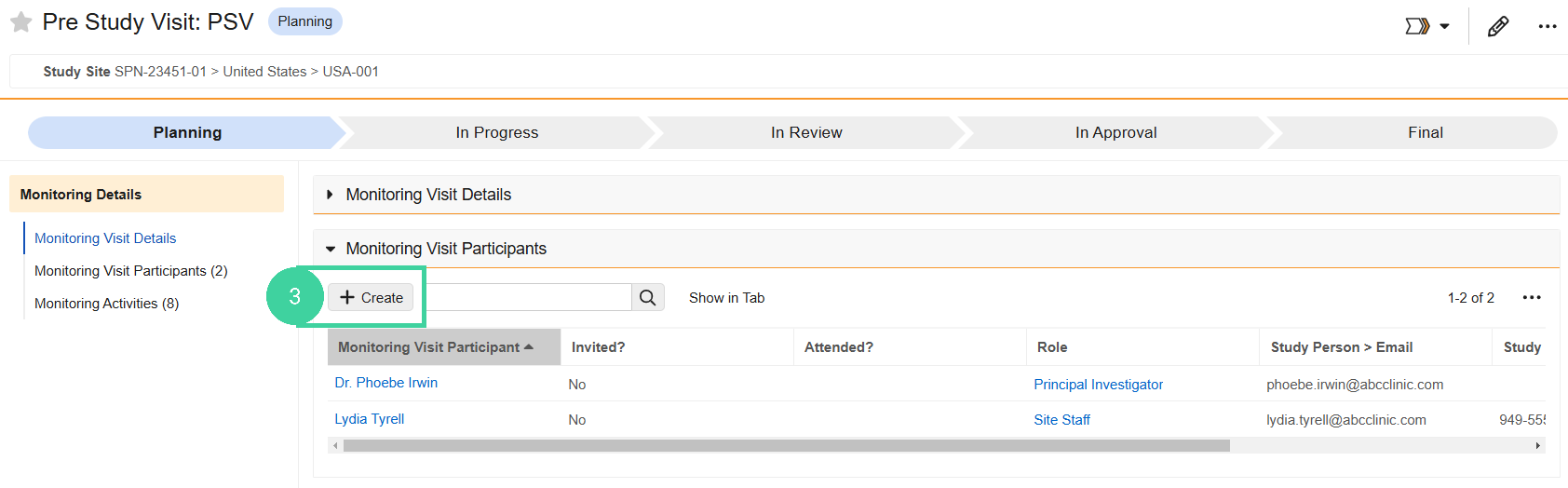
Open the Monitoring Visit Participants section. Site personnel are automatically added as visit participants. Click Create to add additional participants, if needed.
-
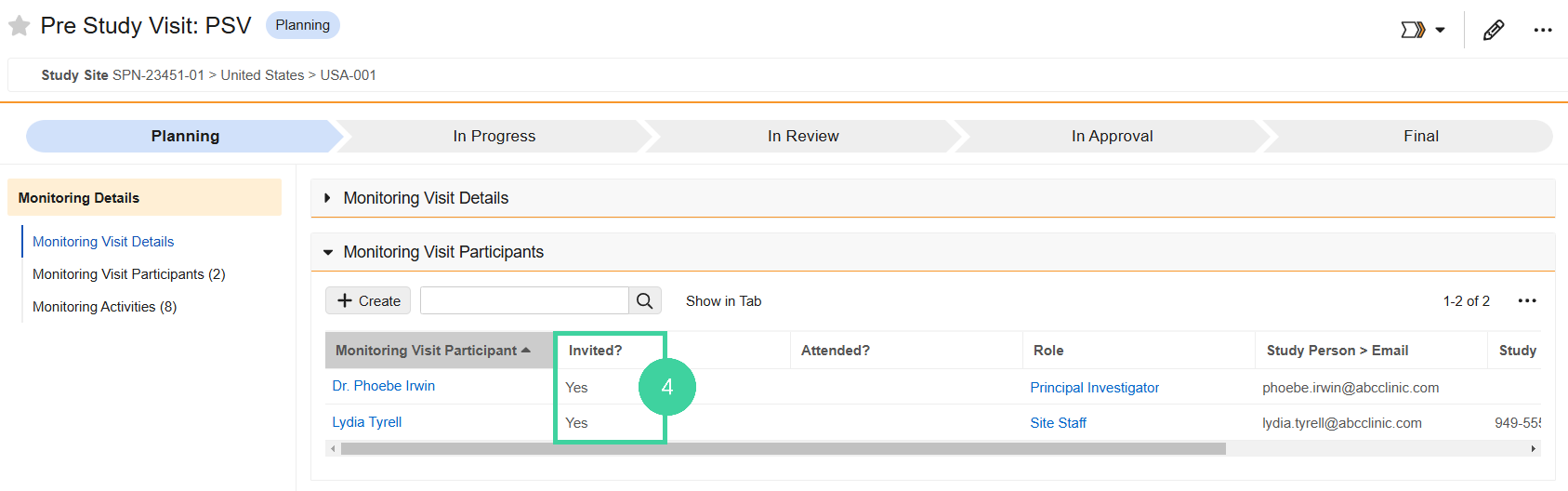
For each participant, set Invited? to Yes if they should be included in the Visit Confirmation letter that gets sent to the site ahead of the visit.
-
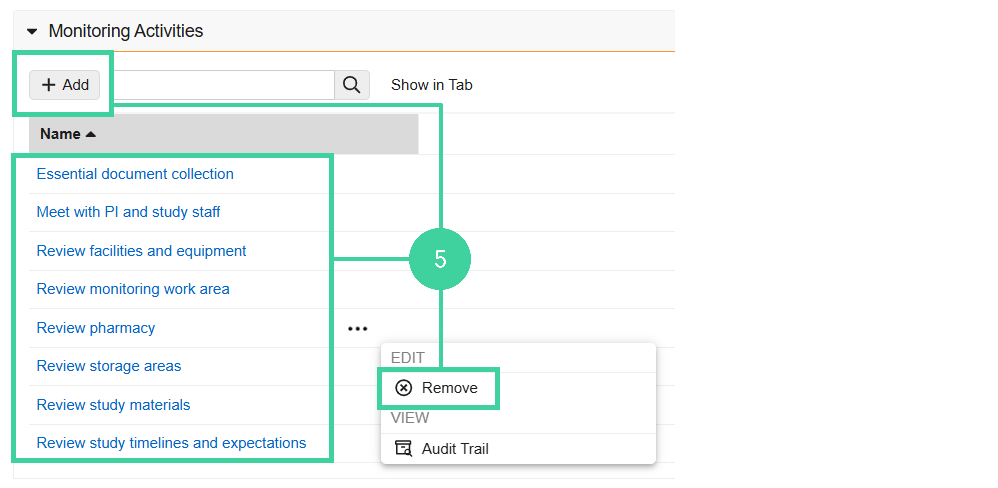
Open the Monitoring Activities section and review the list of activities planned for the visit. The activities will be listed in the Visit Confirmation letter.
-
Click Add to add an additional activity for the visit.
-
Hover over an activity and select Remove from the All Actions menu if it will not be done at this visit.
-
-
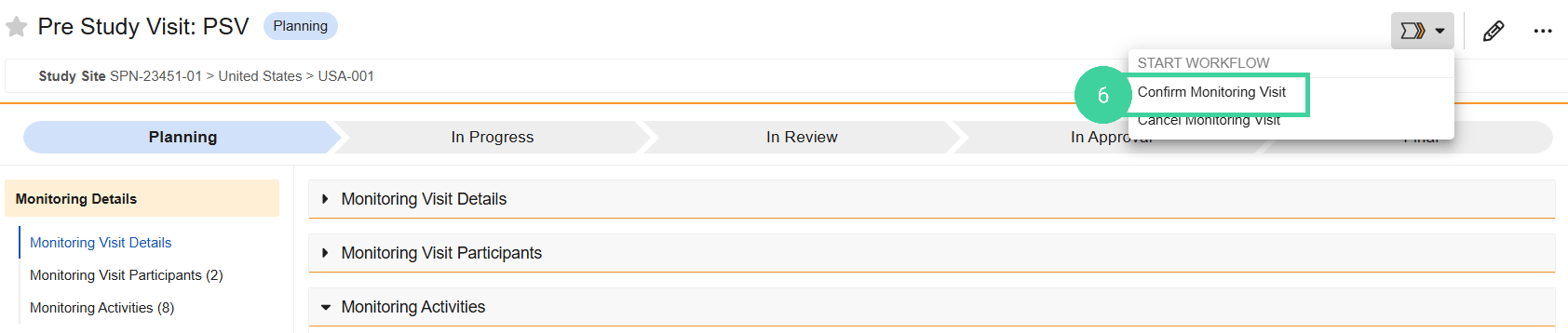
Select Confirm Monitoring Visit from the Workflow Actions menu.
-
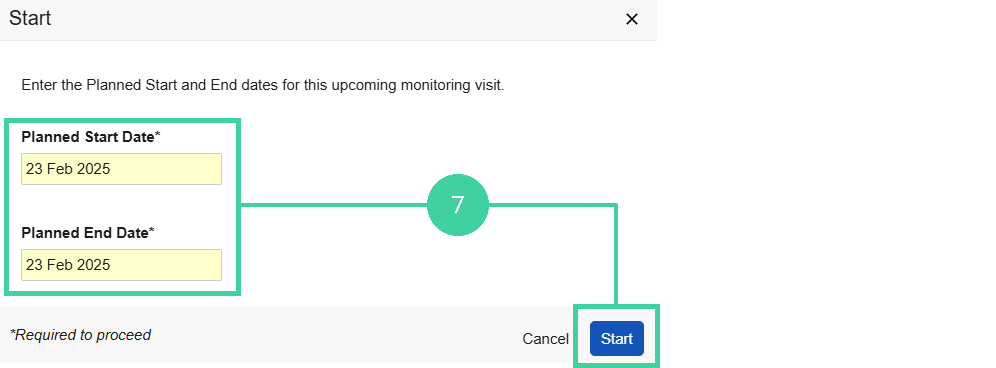
Specify the Planned Start and End Date for the visit and click Start.
What is the next step?