- Clinical Basics: CTMS Vault Admin, Clinical Basics: Study Manager
Visit Definitions are a standard set of visits that subjects can attend during the course of the study, for example, screening visits, baseline visits, treatment visits, or end of trial visits.
If you are connecting your Clinical Basics vault with a Veeva EDC vault, Visit Definitions will be pulled in automatically from Veeva EDC based on the event definitions set up for the study. In this case, you should not create any visit definitions manually.
Note: Visit definitions created at the study level determine the types of subject visits that are able to be created.
To create a visit definition manually:
-
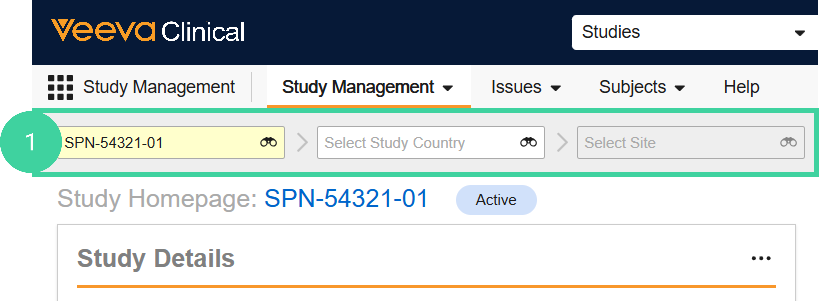
Navigate to the Study Management homepage and select a Study. Do not drill down to the country or site level.
-
Click the study link.
-
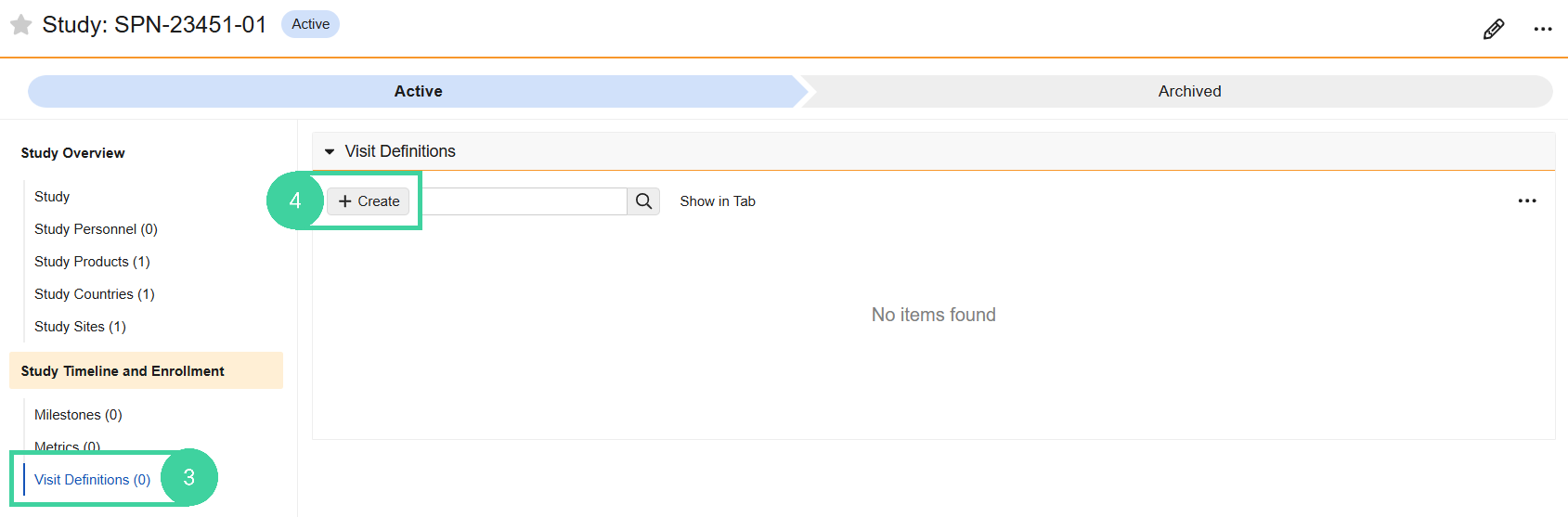
Click Visit Definitions from the Study Timeline and Enrollment group on the left
-
Click Create.
-
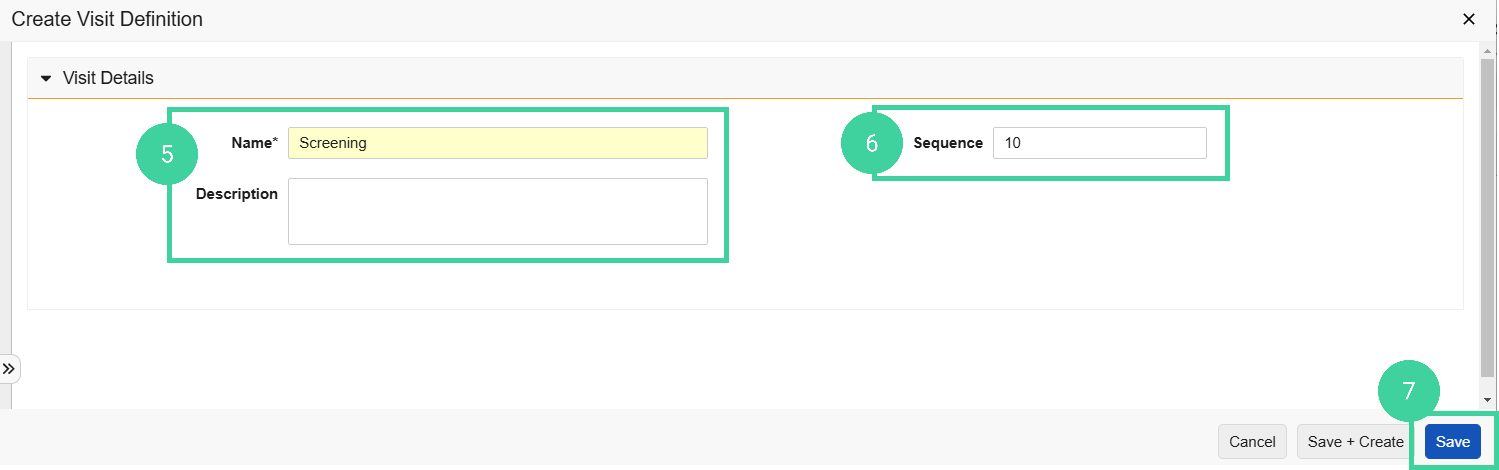
Enter a Name and (optionally) a Description.
-
Optionally, enter a Sequence number.
It can be useful to assign a sequence number to each visit definition so that you can easily sort them in timeline progression, rather than alphabetically. It is a good practice to leave a gap between each sequence number (such as by counts of 10) in case another visit definition needs to be inserted into the study later.
-
Click Save.